 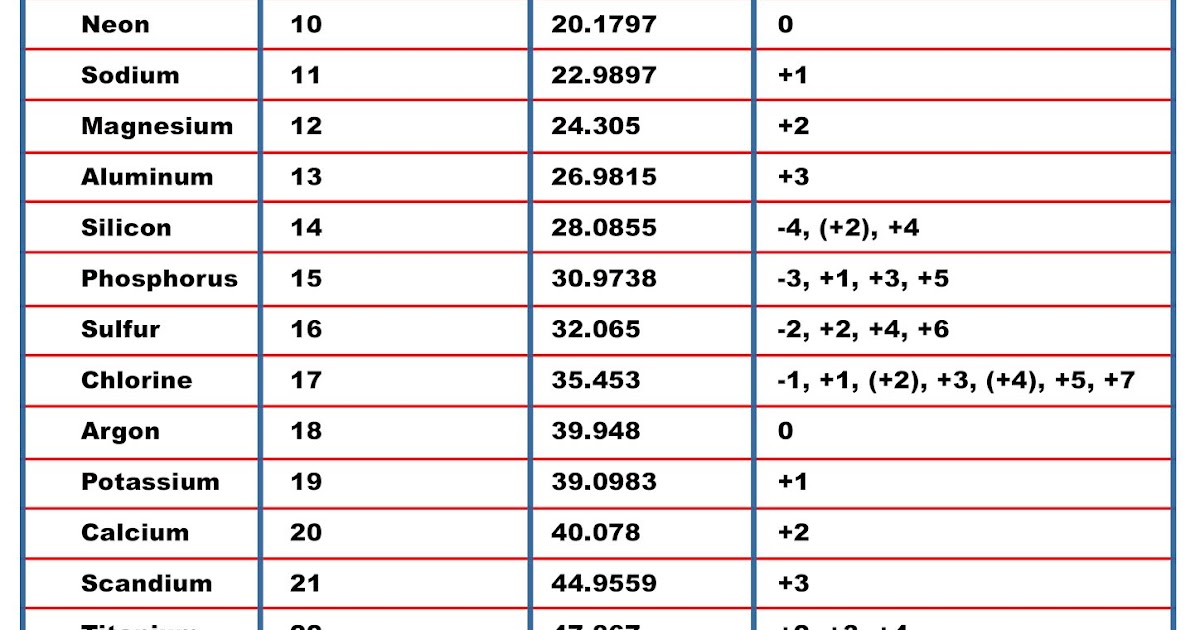 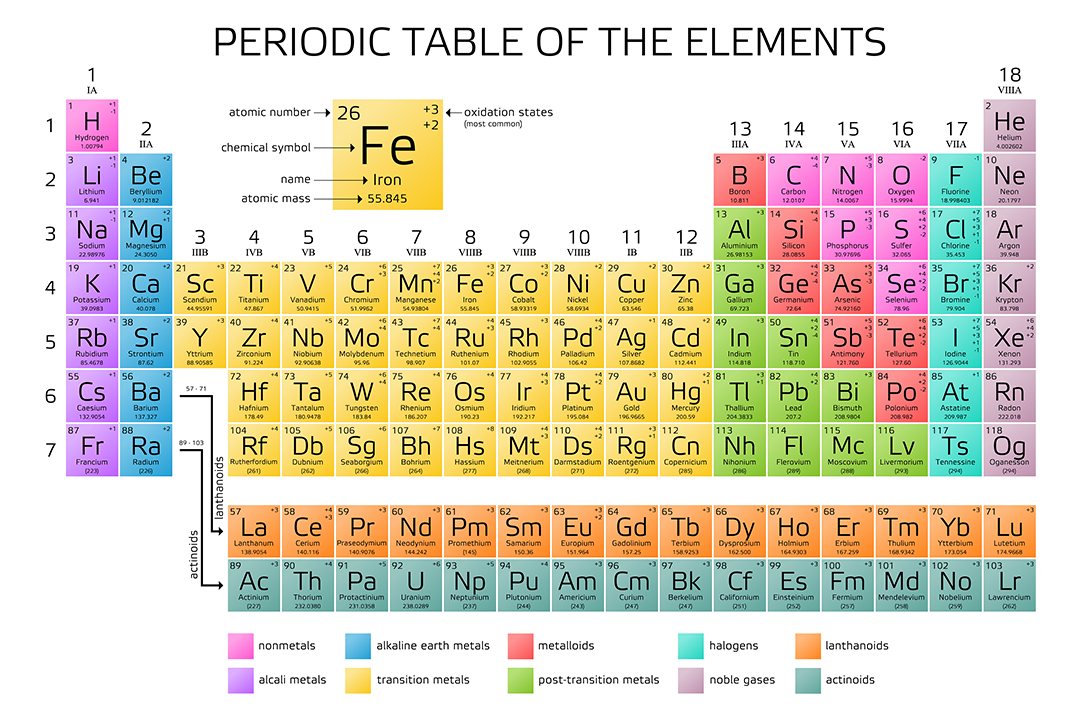 
Sodium has an atomic number of 11 because its nucleus has 11 protons, while oxygen has an atomic number of 8 because its nucleus has 8 protons. X Research source For example, all hydrogen atoms, and only hydrogen atoms, have 1 proton. The atomic number is the number of protons in an element, and never varies. For example, the atomic mass of iron is 55.847 amu, which means one mole of iron atoms would weigh 55.847 grams.įind the atomic number of the element or isotope.However, by simply multiplying an atomic mass by 1 g/mol, a workable quantity is obtained for an element's molar mass - the mass (in grams) of one mole of an element's atoms. Atomic masses, when expressed in amu, as on the periodic table, are technically unitless. Relative atomic masses, as listed on the periodic table, are used to calculate molar masses for atoms and molecules.X Research source Thus, the relative atomic mass listed on the periodic table is suitable as an average value for atoms of a certain element, but not as the mass of a single atom of that element. Chemical elements have different isotopes - chemical forms that differ in mass because of the addition or subtraction of one or more neutrons to the atom's nucleus. Note that the relative atomic masses listed on the periodic table are average values for the associated element.This number is usually expressed as a decimal rather than as a whole number. This is almost always written as a number at the bottom of the element's square on the table, under its one or two letter chemical symbol. Most standard periodic tables list the relative atomic masses (atomic weights) of each element. Atoms are so small, however, that even 500 atoms are too small to see or measure by most common techniques.Locate atomic mass on the periodic table. Sheets of printer paper are packaged in reams of 500, a seemingly large number. 
For example, cans of soda come in a six-pack, eggs are sold by the dozen (12), and pencils often come in a gross (12 dozen, or 144). Many familiar items are sold in numerical quantities with distinct names. The unit that provides this link is the mole (mol), from the Latin moles, meaning “pile” or “heap.” To analyze the transformations that occur between individual atoms or molecules in a chemical reaction, it is therefore essential for chemists to know how many atoms or molecules are contained in a measurable quantity in the laboratory-a given mass of sample. In the laboratory, for example, the masses of compounds and elements used by chemists typically range from milligrams to grams, while in industry, chemicals are bought and sold in kilograms and tons. Because the masses of individual atoms are so minuscule (on the order of 10 −23 g/atom), chemists do not measure the mass of individual atoms or molecules. The problem for Dalton and other early chemists was to discover the quantitative relationship between the number of atoms in a chemical substance and its mass. It also describes the law of multiple proportions, which states that the ratios of the masses of elements that form a series of compounds are small whole numbers. _3(PO_4)_2 = 310.177 \, amu \nonumber \]ĭalton’s theory that each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
